On June 1, 2023, Ashkan Abbey, MD, dosed the first patient in the Phase II SIGLEC Trial, a clinical study evaluating AVD-104, a potential new treatment for geographic atrophy (GA) from age-related macular degeneration (AMD). Developed by Aviceda Therapeutics, AVD-104 is a novel glycan-coated intravitreal nanoparticle designed to alleviate chronic, non-resolving inflammation that can occur with GA.
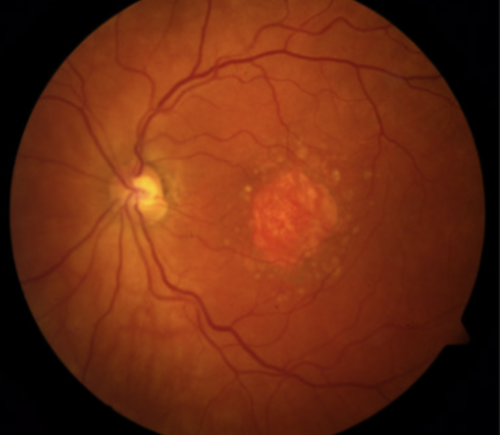
Geographic Atrophy from Macular Degeneration
AMD is a major cause of moderate and severe vision loss in adults over 60 worldwide, currently affecting approximately 11 million people in the United States. It can prominently and permanently reduce central vision, limiting patients’ ability to read, drive and recognize faces.
The wet (neovascular) form of AMD occurs because of abnormal blood vessel growth, bleeding and scarring that destroys central retinal cells. Approved anti-VEGF therapies have helped control and treat this form. The dry (non-neovascular) form of AMD is characterized by the development of GA in which there is irreversible progressive destruction (atrophy) of central retinal cells and underlying blood vessels due to chronic inflammation. Upon examination by an ophthalmologist or retina specialist, these areas of atrophy often look like a map, hence the term “geographic atrophy.”
A New Approach to Treating Geographic Atrophy
AVD-104 was designed to target and attack both of the pathways that cause inflammation in GA:
- Abnormal complement cascade amplification in the retinal photoreceptor, retinal pigment epithelial and choriocapillaris regions in the back of the eye. The body’s complement system (also called complement cascade) is a part of the immune system that enhances the ability of antibodies and phagocytes (a type of white blood cells) to promote inflammation.
- Over-activated macrophage activity resulting in phagocytosis of retinal and retinal pigment epithelial cells. Macrophages are white blood cells that stimulate the action of other immune system cells. Phagocytosis is the process by which phagocytes surround and destroy foreign substances and remove dead cells.
Texas Retina Associates is excited to participate in this new clinical trial.
“I am honored to be the first to dose a new agent that we hope will usher in a generation of safe treatments for GA with better outcomes than were ever possible before,” shares Dr. Abbey.
Click here to learn more about all of the clinical trials currently available at Texas Retina Associates.